|
| Method no.: |
ID-190 (This method
supersedes ID-109) |
| |
|
| Matrix: |
Air |
| |
|
OSHA Permissible Exposure
Limits
Final Rule and
Transitional
Limits: |
25 ppm (Time Weighted
Average) |
| |
|
| Collection
Device: |
The sampling device consists
of:
1) Two glass tubes which contain triethanolamine-impregnated
molecular sieve
2) a middle tube which contains an oxidizer
3)
a personal sampling pump is used to draw a measured volume of air
through the tubes. |
| |
|
| Recommended Sampling
Rate: |
0.025 L/min |
| |
|
| Recommended Maximum Air
Volume: |
6.0 L |
| |
|
| Analytical
Procedure: |
The sample is desorbed using
a 1.5% triethanolamine solution and analyzed as nitrite by ion
chromatography. |
| |
|
Detection Limit
Qualitative:
Quantitative: |
0.11 ppm (6-L air
sample)
0.32 ppm (6-L air sample) |
| |
|
Precision and
Accuracy
Evaluation
Range:
CVT:
Bias:
Overall
Error: |
13.0 to 50.5
ppm
0.082
+3.3%
±19.7% |
| |
|
| Method Classification: |
Validated Method |
| |
|
| Chemist: |
James Ku |
| |
|
| Date (Date Revised): |
April, 1989 (May,
1991) |
| |
|
Commercial manufacturers and products mentioned in this method are
for
descriptive use only and do not constitute endorsements by
USDOL-OSHA.
Similar products from other sources can be
substituted.
|
| |
|
Branch of Inorganic Methods Development
OSHA Technical
Center
Sandy, Utah
|
1.
Introduction
This method
describes the collection and analysis of airborne nitric oxide (NO).
Samples are taken in the breathing zone of workplace personnel and
analyses are performed by ion chromatography (IC).
1.1. History
Previous methods involved
oxidation of NO to nitrogen dioxide (NO2) using a
chromate compound and subsequent conversion of NO2 to
nitrite using triethanolamine-impregnated molecular sieve
(TEA-IMS) sampling tubes. Common methods used a combination
sampling tube and NO was determined colorimetrically (as
NO2-) using a modified Griess-Saltzman
reaction (8.1.-8.2.). This method, like most colorimetric
procedures, can have significant interferences.
A
differential pulse polarographic (DPP) method (8.3.) was later
developed to improve analytical sensitivity and decrease the
potential for interferences. The sensitivity of the DPP method was
more than adequate for measuring workplace concentrations of NO;
however, the nitrite ion is unstable in the pH range (pH 1-2) used
during analysis (8.4.).
Method no. ID-190 uses the TEA-IMS
sampling tube/chromate oxidizer approach. Samples are analyzed by
IC.
1.2. Principle
A known volume of air is drawn
through the sampling device which captures any nitrogen dioxide
(NO2) in the sampled air and also converts any NO to
nitrite ion (NO2-). The sampling device
consists of three glass tubes connected in series. The front and
back tubes contain TEA-IMS, the middle or oxidizer tube contains
an inert carrier impregnated with a chromate salt. The first
TEA-IMS tube does not capture NO; this tube is only used to
capture and convert to NO2- any
NO2 present in the sampled air. The middle tube
oxidizes the sampled NO to NO2. The back TEA-IMS tube
then captures and converts this NO2 to
NO2-. Both TEA-IMS samples are desorbed
using an aqueous triethanolamine (TEA) solution and analyzed as
NO2- by IC. The front tube analytical
results are reported as NO2 and the back tube as NO.
The conversion mechanism of NO2 gas to
NO2- has been proposed by Gold (8.5.). The
following is Gold's proposal for the reaction of equivalent
amounts of NO2 and TEA in an aqueous solution:
2NO2<=>
N2O4
N2O4 +
(HOCH2CH2)3N -->
(HOCH2CH2)3NNO+NO3-
(HOCH2CH2)3NNO+NO3-
+ H2O -->
(HOCH2CH2)3NH+NO3-
+ HNO2
HNO2 -->
H+ + NO2-
|
Nitrogen dioxide disproportionates to
NO2- and nitrate
(NO3-) in the presence of TEA and water. The
NO2- formed from the above reaction can be
analyzed via conventional analytical methods (8.1.-8.4.,
8.6.-8.7.) including IC. Unfortunately NO3-
is found in the commercial TEA-IMS sorbent as a significant
contaminant. This contamination ruled out further research to also
measure this NO2-TEA disproportionation product by IC.
This reaction path requires a stoichiometric factor of 0.5
for the conversion of
gaseous NO2 to
NO2-. Experiments indicate the
stoichiometric factor of 0.5 is seen only when NO2
concentrations are greater than 10 ppm (8.5., 8.8.-8.9.). The
conversion factor has been experimentally determined to average
approximately 0.6 to 0.7 when concentrations are below 10 ppm
(8.1.-8.3., 8.5.-8.9.). The deviation from ideal stoichiometry is
believed to be due to competing reactions; however, evidence to
support a competing mechanism has not been found (8.5.).
1.3. Advantages and Disadvantages
1.3.1. This method has adequate sensitivity for
determining compliance with the OSHA Time Weighted Average (TWA)
Permissible Exposure Limit (PEL) for workplace exposures to NO.
1.3.2. The sampling device can be used to simultaneously
collect NO and NO2; however, results for
NO2 may not reflect short-term exposures (see Section
5.2. for more details).
1.3.3. The analysis is simple,
rapid, easily automated and is specific for
NO2-.
1.3.4. After analytical
sample preparation, NO exposures (as nitrite ion) can also be
determined by colorimetric or polarographic analytical
techniques (8.1.-8.3.).
1.3.5. A disadvantage is the
potential interference from large amounts of soluble chloride
salts present in commercial molecular sieve. Prior to TEA
impregnation, the molecular sieve should be washed with
deionized water (DI H2O) to remove any soluble
chloride salts.
1.3.6. Another disadvantage is the need
for a concentration-dependent conversion factor when calculating
results. 1.4. Physical properties (8.10., 8.11.)
Nitric oxide (CAS No. 10102-43-9), one of several oxides
of nitrogen, is a colorless gas. A deep blue color is usually
noted when NO is in the liquid state and a bluish-white color when
solid. Other physical characteristics of NO are:
| Formula weight |
30.01 |
| Specific gravity |
1.27 at -150.2 °C (as
liquid) |
| Melting point |
-163.6 °C |
| Boiling point |
-151.8 °C |
| Vapor density |
1.04 (air = 1) |
| Solubility |
4.6 mL NO in 100 mL H2O |
| Synonyms |
nitrogen monoxide*, |
| |
mononitrogen
monoxide | *Nitrogen monoxide has also been used as a synonym
for nitrous oxide (N2O).
1.5. Some industrial
sources for potential nitric oxide exposures are:
agricultural silos
arc or gas welding (esp.
confined space operations)
electroplating plants
food and
textile bleaching
jewelry manufacturing
metal nitrosyl
carbonyl production
nitric acid production
nitrogen
fertilizer production
nitro-explosive production
nitrosyl
halide production
pickling plants Nitrogen dioxide
and nitric oxide usually exist together in industrial settings.
Nitric oxide is reactive in air and produces NO2
according to the following equations (8.10.):
2NO +
O2 ----> 2NO2
d(NO2)/dt=
K(O2)(NO)2 |
(K is a temperature dependent constant. At 20 °C, K
= 14.8 X 109)
An experimental approximation of
the NO/NO2 distribution found in various industrial
operations is shown (8.10.):
| Source |
%
NO2 |
%
NO |
|
| Carbon arc |
9 |
91 |
| Oxyacetylene torch |
8 |
92 |
| Cellulose nitrate combustion |
19 |
81 |
| Diesel exhaust |
35 |
65 |
| Dynamite blast |
52 |
48 |
| Acid dipping |
78 |
22 |
The potential for exposure to both NO2
and NO should be considered because NO is easily oxidized to
NO2 and both oxides are likely to coexist in industrial
settings.
1.6. Toxicology (8.11.-8.14.)
Information listed within this section is a synopsis of
current knowledge of the physiological effects of nitric oxide and
is not intended to be used as a basis for OSHA policy.
1.6.1. Nitric oxide is classified as a respiratory
irritant. The main route of exposure is inhalation; however,
physiological damage can also occur eyes or skin.
The
term "silo-fillers' disease" has been used to describe exposure
to nitric as well as other nitrogen oxides. The national
population-at-risk for exposure to nitrogen oxides has been
estimated by NIOSH to be approximately 950,000 employees
(National Occupational Hazard Survey, 1972-74). When
encountering either NO or NO2 at high concentrations,
both species will usually be present. Little scientific data is
available regarding exposures to NO only. The majority of
collected data concerns exposure to NO2 since NO
appears to be only one-fifth as toxic as NO2 at low
concentrations. Symptoms immediately following NO exposure are
usually mild or not apparent. Severe symptoms may not appear up
to 72 hours after exposure.
1.6.2. Mild exposures to NO
can result in symptoms such as:
| cough |
shortness of
breath |
| painful breathing |
chest pains |
| increased breathing rate |
weakness |
| methemoglobinemia |
|
More severe exposures (>100 ppm) are
characterized by pulmonary edema , cyanosis, pneumonia, severe
methemoglobinemia, respiratory failure, and death.
1.6.3. The IDLH (Immediately Dangerous to Life or
Health) concentration is 100 ppm NO. The LCLo (Lethal
Concentration - Low) for inhalation by mice is 320 ppm.
1.6.4.Mechanism for toxicity:
Nitric oxide is
slightly soluble in water and forms nitrous and nitric acid.
This reaction occurs with lung tissue and produces respiratory
irritation and edema. Alkali present in the lung tissue
neutralizes the nitrous and nitric acids to nitrite and nitrate
salts which are then absorbed into the bloodstream. The end
result is the formation of nitroxy-hemoglobin complexes and
methemoglobin in the circulatory system.
The formation
of hemoglobin complexes is thought to contribute to the toxicity
of NO but is not considered to be the sole source of the toxic
reaction. The respiratory damage from nitrous and nitric acid
appears to be more significant.
2. Range,
Detection Limit and Sensitivity
The analytical parameters and limits of this method
have been previously described (8.8.). Brief descriptions are
provided in Section 3 below.
3. Method
Performance
This method was evaluated in the
concentration range of 13.0 to 50.5 ppm. Air volumes of
approximately 6 L and flow rates of about 0.025 L/min were used.
Samples were collected for 240 min. Sample results were calculated
using the concentration-dependent conversion factors mentioned in
Section 7. Listed on the cover page (CVT, bias, overall
error) and below are evaluation data taken from the backup report
(8.9.).
|
| Qualitative detection
limit1: |
0.08 µg/mL (as
NO2-) |
|
0.11 ppm NO (6 L air
volume) |
| Quantitative detection
limit1: |
0.23 µg/mL (as
NO2-) |
|
0.32 ppm NO (6 L air
volume) |
| Sensitivity (1 to 30 µg/mL
nitrite): |
|
| Hewlett-Packard2 |
239,000 area counts per |
|
1 µg/mL
NO2- |
| Dionex2 |
10,000 area counts per |
|
1 µg/mL
NO2- |
| Collection
efficiency3 |
100% |
| Breakthrough |
none at levels
tested3 |
| Sample storage |
at least 30 days (20-25 °C) |
|
| 1 |
Detector setting = 3
microsiemens, sample loop = 50 µL (8.8.) |
| 2 |
A model 3357 data
reduction system (Hewlett-Packard, Avondale, PA) (1 area unit
= 0.25 microvolt-second) was used during first part of
evaluation. An AutoIon 400 data reduction system (Dionex,
Sunnyvale, CA) was used for later analyses. |
| 3 |
Collection efficiency
samples were taken using a concentration of 50.5 ppm NO for
240 min, 50% RH, and 0.025 L/min. Breakthrough tests were
performed at 25 °C, 50% RH, and a flow rate of 0.025 L/min.
Samples were collected at a concentration of 200 ppm for 60,
120, 180, and 240 min. |
4. Interferences
4.1. When other compounds are
known or suspected to be present in the sampled air, such
information should be transmitted to the laboratory with the
sample.
4.2. Any compound that has the same retention time
as nitrite, when using the operating conditions described, is an
interference.
4.3. Interferences may be minimized by
changing the eluent concentration, column characteristics, and/or
pump flow rate.
4.4. If there is an unresolvable
interference, alternate polarographic or colorimetric methods may
be used (8.1.-8.3.).
4.5. Contaminant anions normally
found in molecular sieve, such as NO3-,
SO42-, and PO43-, do
not interfere. Large amounts (greater than 4 to 5 µg/mL) of
Cl- can interfere. 5. Sampling
5.1. Equipment
5.1.1. A three tube sampling device is commercially
available (NO/NO2 sampling tubes, Cat. No.
226-40-special order, water-washed, SKC,
Eighty Four, PA) and can be used to simultaneously sample
NO2 and NO, or sample only NO2. This
device consists of three flame-sealed glass tubes:
1) Nitrogen dioxide is collected in the first tube
which contains 400 mg TEA-IMS.
2) The second (oxidizer)
tube converts NO to NO2 and contains approximately
1 g of a chromate compound impregnated on an inert carrier.
3) The last 400 mg TEA-IMS packed tube collects the
converted NO2. All
molecular sieve used for tube packing must be washed with
DI H2O before impregnation with TEA. The dimensions
of each TEA-IMS tube are 7-mm o.d., 5-mm i.d., and 70-mm long. A
3-mm portion of silylated glass wool is placed in the front and
rear of each tube. The dimensions of the oxidizer tube are 7-mm
o.d., 5-mm i.d., and 110-mm long .
When the three tubes
are connected in series as shown below, NO2 and NO
can be collected simultaneously. The first TEA-IMS tube must be in place to prevent the collection of
NO2 by the second TEA-IMS tube.
THREE-TUBE SAMPLING DEVICE
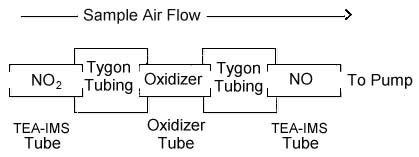
|
| Text
Version: The first tube in the Three-Tube Sampling Device
is a nitrogen dioxide (NO2) sampling tube
(TEA-IMS Tube). The second tube in the series is an
oxidizer tube, and the third is another NO2
sampling tube that is identical to the first tube. The
three tubes are connected with short lengths of plastic
tubing (Tygon or equivalent). The three tubes should be
connected as close to one another as possible. The
sampling device is connected to the sampling pump with
flexible plastic tubing. The set of three tubes that
compose the sampling device is available from SKC, Inc. as
catalog 226-40. |
5.1.2. Personal sampling pumps capable
of sampling at a flow rate of approximately 0.025 L/min are
used.
5.1.3. A stopwatch and bubble tube or meter are
used to calibrate pumps. A sampling device is placed in-line
during flow rate calibration.
5.1.4. Various lengths of
Tygon tubing are used to connect the sampling tubes and pump
together. 5.2. Sampling
Procedure
Note: If sampling for both NO2 and NO is necessary,
two separate pumps and sampling devices should be used. The
differences in OSHA exposure limits [the NO2 PEL is a 1
ppm Short-Term Exposure Limit (8.15.). Nitric oxide is a TWA PEL.]
and flow rates dictates a need for a separate assessment of
NO2. Nitric oxide is collected at a 0.025 L/min pump
flow rate. Nitrogen dioxide can be collected at this flow rate;
however, a longer sampling time will be necessary to collect a
detectable amount of NO2 than for a short-term
measurement. Concentrations of NO2 may vary in the
workplace during a longer sampling period.
5.2.1. Calibrate the sampling pumps to a flow rate
of 0.025 L/min.
5.2.2. Connect the sampling device to
the pump. The different sampling schemes are listed:
a) Sampling for NO2
only: - Use a single TEA-IMS tube (8.8.).
b) Sampling for both NO and NO2: The
three-tube device is used. The device must be assembled as
shown above.
Label the first tube
"NO2".
Label the tube following the oxidizer
section "NO". 5.2.3. Place the sampling tube or
device in the breathing zone of the employee.
5.2.4.
Collect the sample at the listed flow rates and sampling times:
a) For NO2 only:
0.200 L/min for at least 15 min (8.8.) per sample.
b) For both NO and NO2: 0.025 L/min
for 4 h per sample (Note: The front ube of the three-tube device
can be submitted for NO2 analysis; however,
analytical results may not represent short-term exposures).
5.2.5. The maximum recommended air volume is 6 L per NO
sample.
Take enough samples for NO to cover the
workshift.
Note: One oxidizer tube per sample is sufficient for
concentration ranges of NO usually encountered in industrial
settings. A color change from orange to blue-green will be
noticeable if the oxidizer is depleted.
6. Analysis
6.1. Precautions
6.1.1. Refer to instrument and standard operating
procedure (SOP) (8.16.) manuals for proper operation.
6.1.2. Observe laboratory safety regulations and
practices.
6.1.3. Sulfuric acid
(H2SO4) can cause severe burns. Wear
protective eyewear, gloves, and labcoat when using concentrated
H2SO4. 6.2. Equipment
6.2.1. Ion chromatograph (Model 2010 or 4000,
Dionex, Sunnyvale, CA) equipped with a conductivity detector.
6.2.2. Automatic sampler (Model AS-1, Dionex) and 0.5 mL
sample vials.
6.2.3. Laboratory automation system: Ion
chromatograph interfaced to a data reduction and control system
(AutoIon 400 or 450, Dionex).
6.2.4. Micromembrane
suppressor, anion (Model AMMS-1, Dionex).
6.2.5.
Separator and guard columns, anion (Model HPIC-AS4A and AG4A,
Dionex).
6.2.6. Disposable syringes (1 mL) and filters.
Note: Some syringe pre-filters are not cation- or
anion-free. Tests should be done with blank solutions first to
determine suitability for the analyte being determined.
6.2.7. Erlenmeyer flasks, 25-mL, or scintillation vials,
20-mL.
6.2.8. Miscellaneous volumetric glassware:
Micropipettes, volumetric flasks, graduated cylinders, and
beakers.
6.2.9. Analytical balance (0.01 mg).
6.3. Reagents - All chemicals should be at least
reagent grade.
6.3.1. Deionized water (DI H2O) with a
specific conductance of less than 10 microsiemens.
6.3.2. Triethanolamine
[(HOCH2CH2)3N]
sodium
carbonate (Na2CO3)
sodium bicarbonate
(NaHCO3)
sulfuric acid
(H2SO4, concentrated 95 to 98%)
sodium
nitrite (NaNO2)
6.3.3. Liquid desorber (1.5%
TEA):
Dissolve 15 g TEA in a 1-L volumetric flask which
contains approximately 500 mL DI H2O. Add 0.5 mL
n-butanol and then dilute to volume with DI H2O.
6.3.4. Eluent (2.0 mM Na2CO3/1.0
mM NaHCO3):
Dissolve 0.848 g
Na2CO3 and 0.336 g NaHCO3 in
4.0 L DI H2O.
6.3.5. Regeneration solution
(0.02 N H2SO4):
Place 1.14 mL
concentrated H2SO4 into a 2-L volumetric
flask which contains about 500 mL DI H2O. Dilute to
volume with DI H2O.
6.3.6. Nitrite stock
standard (1,000 µg/mL):
Dissolve 1.5000 g NaNO2
and dilute to the mark with DI H2O in a 1-L
volumetric flask. Prepare every three months.
6.3.7.
Nitrite standard (100 µg/mL):
Dilute 10 mL of 1,000 µg/mL
nitrite stock standard to 100 mL with liquid desorber. Prepare
monthly.
6.3.8. Nitrite standard (10 µg/mL):
Dilute
10 mL of 100 µg/mL nitrite stock standard to 100 mL with liquid
desorber. Prepare weekly.
6.3.9. Nitrite standard (1
µg/mL):
Dilute 10 mL of 10 µg/mL nitrite stock standard to
100 mL with liquid desorber. Prepare daily. 6.4.
Working Standard Preparation
6.4.1. Nitrite working standards (10-mL final
volumes) may be prepared in the ranges specified below:
| Working Std |
Standard |
Aliquot |
µg/mL
|
Solution µg/mL
|
mL
|
| 0.5 |
1 |
5 |
| 1 |
1 |
* |
| 3 |
10 |
3 |
| 6 |
10 |
6 |
| 10 |
10 |
* |
| 30 |
100 |
3 |
| 50 |
100 |
5 |
* Already prepared in Section 6.3.
6.4.2.
Pipette appropriate aliquots of standard solutions (prepared in
Section 6.3.) into 10-mL volumetric flasks and dilute to volume
with liquid desorber.
6.4.3. Pipette a 0.5- to 0.6-mL
portion of each standard solution into separate automatic
sampler vials. Place a 0.5-mL filter cap into each vial. The
large exposed filter portion of the cap should face the standard
solution.
6.4.4. Prepare a reagent blank from the liquid
desorber solution. 6.5.
Sample Preparation
6.5.1. Identify which tube is the collected
NO2 sample and which is NO. Analyze these two tubes
as separate samples.
6.5.2. Discard the oxidizer tube
appropriately. This tube contains a chromate salt and may be
considered a hazardous waste. Local regulations or restrictions
should be consulted before disposal.
6.5.3. Clean the
25-mL Erlenmeyer flasks or scintillation vials by rinsing with
DI H2O.
6.5.4. Carefully remove the glass
wool plugs from the sample tubes, making sure no sorbent is lost
in the process. Transfer each TEA-IMS section to individually
labeled 25-mL Erlenmeyer flasks or scintillation vials.
6.5.5. Add 10 mL of liquid desorber to each flask
containing NO samples, shake vigorously for about 30 s. Allow
the solution to stand for at least 1 h. (Note: Add 3 mL to
NO2 samples - see reference 8.8. for further details
regarding NO2 analysis and result calculations)
6.5.6. If the sample solutions contain suspended
particulate, remove the particles using a pre-filter and
syringe. Fill the 0.5-mL automatic sampler vials with sample
solutions and push a filtercap into each vial. Label the vials.
6.5.7. Load the automatic sampler with labeled samples,
standards and blanks. 6.6. Analytical Procedure
Set up the ion chromatography and analyze the samples and
standards in accordance with the SOP (8.16.). Typical operating
conditions for equipment mentioned in Section 6.2. are listed
below.
| Ion chromatograph |
|
| Eluent: |
2.0 mM
Na2CO3/1.0 mM
NaHCO3 |
| Column temperature: |
ambient |
| Sample injection loop: |
50 µL |
|
|
| Pump |
|
| Pump pressure: |
approximately 1,000 psi |
| Flow rate: |
2 mL/min |
|
|
| Chromatogram |
|
| Run time: |
6 min |
| Average retention time: |
approximately 2
min | 7. Calculations
7.1. Obtain hard copies of
chromatograms from a printer. A typical chromatogram is shown in
Figure 1.
7.2. Prepare a concentration-response curve by
plotting the concentration of the standards in µg/mL (or µg/sample
if the same volumes are used for samples and standards) versus
peak areas or peak heights. Calculate sample concentrations from
the curve and blank correct all samples.
7.3. The
concentration of NO in each air sample is expressed in ppm and is
calculated as:
| ppm NO = |
MV × µg/mL
NO2¯ × solution volume × conversion × GF
formula weight × air volume |
Where:
| MV (Molar
Volume) |
= |
24.45 (25 °C and
760 mmHg) |
| µg/mL NO2 |
= |
blank corrected sample
result |
| Conversion [NO2
(gas)/NO2-] |
= |
varies with
concentration* |
| GF (Gravimetric factor
NO/NO2) |
= |
0.6522 |
| Formula Weight (NO) |
= |
30.01 |
*The conversion of gaseous NO2 to
NO2- is concentration dependent. The final
concentration of NO should be calculated using whichever example
given below is appropriate:
Below 10 ppm NO
From 0 to 10 ppm, the average ratio has been
experimentally determined to be (8.1.-8.3., 8.5.-8.9.):
1 µg NO2 (gas) = 0.630 µg
NO2-
or conversely:
1
µg NO2- = 1.587 µg NO2
(gas)
Simplifying the equation and calculating the
ppm NO using a 10-mL sample volume gives:
| ppm NO =
|
µg/mL
NO2¯ × 10 mL × 0.843
air volume, L |
Above 10 ppm NO
Above 10 ppm NO,
the expected stoichiometric factor of 0.5 mole of nitrite to
1 mole of nitrogen dioxide gas is seen (8.5., 8.8.-8.9.).
Therefore, the following calculation should be used for
sample results above 10 ppm and a
10-mL solution volume:
| ppm NO =
|
µg/mL
NO2¯ × 10 mL × 1.063
air volume,
L | |
7.4. Reporting Results
Report all results to
the industrial hygienist as ppm nitric oxide.
8.
References
8.1. National Institute for
Occupational Safety and Health (NIOSH): NIOSH Manual of Analytical Methods, 2nd ed.,
Vol. 4 (DHEW/NIOSH Pub. No. 78-175, Method No. S321).
Cincinnati, OH. 1978.
8.2. Willey, M.A.,
C.S. McCammon, Jr., and L.J. Doemeny: A Solid Sorbent
Personal Sampling Method for the Simultaneous Collection of
Nitrogen Dioxide and Nitric Oxide in Air. Am.
Ind. Hyg. Assoc. J. 38:358-363 (1977).
8.3.
Occupational Safety and Health Administration
Analytical Laboratory: OSHA Analytical
Methods Manual (USDOL/OSHA-SLCAL Method No. ID-109).
Cincinnati, OH: American Conference of Governmental Industrial
Hygienists (Pub. No. ISBN: 0-936712-66-X), 1985.
8.4. Chang, S.K., R. Kozenianskas, and
G.W. Harrington: Determination of Nitrite Ion Using
Differential Pulse Polarography. Anal.
Chem. 49: 2272-2275
(1977).
8.5. Gold, A.:
Stoichiometry of Nitrogen Dioxide Determination in Triethanolamine
Trapping Solution. Anal. Chem. 49:1448-50 (1977).
8.6. Blacker, J.H.: Triethanolamine for Collecting
Nitrogen Dioxide in the TLV Range. Am. Ind.
Hyg. Assoc. J. 34:390 (1973).
8.7. Saltzman, B.E.: Colorimetric
Microdetermination of Nitrogen Dioxide in the Atmosphere. Anal. Chem. 26:1949
(1954).
8.8. Occupational Safety and
Health Administration Technical Center: Determination of Nitrogen Dioxide in Workplace
Atmospheres (Ion Chromatography) by J.C. Ku
(USDOL/OSHA-SLTC Method No. ID-182).
Salt Lake City UT. Revised 1991.
8.9. Occupational Safety and Health Administration Technical
Center: Nitric Oxide Backup Data Report
(ID-190) by J.C. Ku. Salt Lake City, UT. Revised 1991.
8.10. National Institute for Occupational
Safety and Health: Criteria for a
Recommended Standard...Occupational Exposure to Oxides of Nitrogen
(Nitrogen Dioxide and Nitric Oxide) (DHEW/NIOSH Pub. No.
76-149). Cincinnati, OH: NIOSH, 1976.
8.11. Braker, W. and A.L. Mossman: Matheson Gas Data Book. 5th ed. East
Rutherford, NJ: Matheson Gas Products, 1971. pp.
405-410.
8.12. Merchant,
J.A. Ed.: Occupational Respiratory
Diseases (DHHS/NIOSH Pub. No. 86-102). Cincinnati, OH:
NIOSH, 1986. pp. 590-594.
8.13. American Conference of Governmental Industrial
Hygienists: Documentation of the
Threshold Limit Values and Biological Exposure Indices. 5th
ed. Cincinnati, OH: ACGIH, 1986. pp. 435-436.
8.14. Specialty Gas Department:
Material Safety Data Sheet for Nitric Oxide. Allentown, PA: Air
Products, 1982.
8.15. "Air Contaminants; Final Rule":
Federal Register 54:12 (19 Jan. 1989).
pp. 2521-2523.
8.16. Occupational Safety and Health Administration Technical
Center: Standard Operating Procedure -
Ion Chromatography. Salt Lake City, UT. In progress
(unpublished). |
| |
| |

